


 |
 |
 |
| Home | Initium | Angiogenesis | FAQ |
| Endothelial Contribution to Angiogenesis |
|---|
|
Graphical overview showing the involvement of TEMs in cellular processes 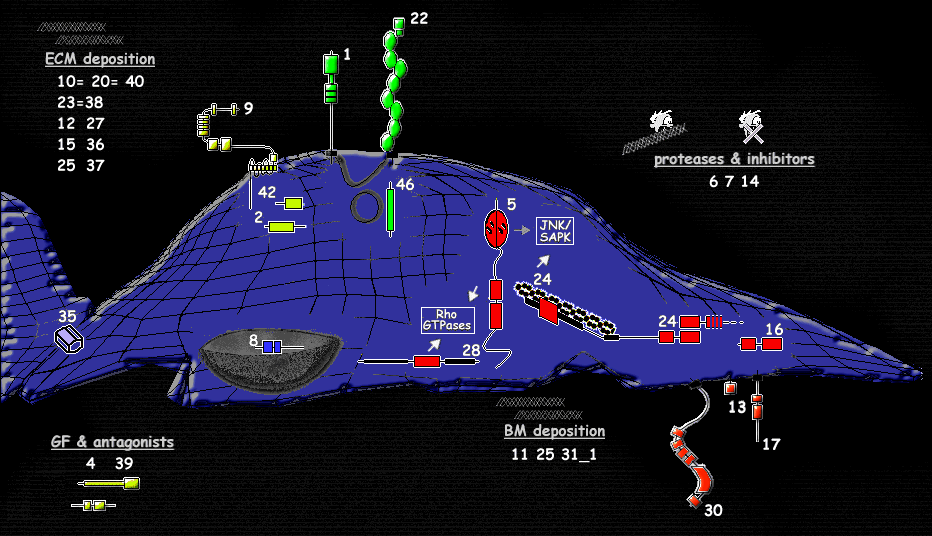 |
|
This sequence-analytic study shows that the endothelial response to the angiogenic stimuli released by a tumor contibutes to the following cellular processes controlling angiogenesis:
|
Thus, the changes in endothelial mRNA expression are responsible for the executive steps of angiogensis including (extracellular matrix) ECM destruction, migration, control of new ECM deposition. Limited insight could be gained about angiogenesis initiation and control. The experimental approach implies that the expression profile represents an average of endothelial cells at different stages of angiogenesis.
| INVOLVEMENT OF TEMs IN POLICING STEPS OF ANGIOGENESIS | |||
| general context signaling - extracellular signaling factors:
|
Growth factors are involved in all stages of angiogenesis and induce collagen deposition, cellular proliferation, migration and morphogenetic events.
In the particular context it is interesting that: 1) the transforming growth factor-beta system, which leads to expression of matrix components and enhanced proteolytic activity, is regulated by members of the steroid-thyroid hormone superfamily; 2) Dkk-3 is potentially a thyroid hormone-binder involved in the modulation of thyroid hormone action; 3) TEM2 might show thyroid hormone-dependent expression. TEM9 is G-protein coupled receptor for a yet unknown peptide/protein. TEM42 is a G-protein signaling regulator; thus, a functional link between the coexpressed TEM9 and TEM42 cannot be excluded. | ||
cell-cell communication
|
Gap-junctional intercellular coupling is necessary for the coordination of endothelial cells during angiogenesis. The reconstitution of cell-cell communications is of particular importance in the final steps of regeneration of the vascular network. | ||
transcriptional regulation
|
A function for Hairy/E(SPL) transcription factors in angiogenesis is well established. | ||
| INVOLVEMENT OF TEMs IN EXECUTIVE STEPS OF ANGIOGENESIS AND THEIR REGULATION | |||
| ECM- & BM- deposition and remodeling - proteases and inhibitors
|
ECM- & BM- deposition and remodeling in the course of angiogenesis 1. In early angiogenesis activated endothelial cells penetrate the basement membrane and invade the ECM surrounding the existing vessel. Proteases, such as matrix metalloproteases, mediate the degradation of ECM components, activate latent growth factors or release them from their ECM binding sites. 2. A step of chemotactic migration follows. 3. Molecular coupling events are followed by lumen formation, basement membrane generation, pericyte recruitment and vessel maturation. In late angiogenesis the basement membrane is tightly bound to cells comprising the vessel wall, provides inductive signals, and plays a important role in the homeostasis of newly formed vessels. Basal lamina deposition is potentially implicated in early as well as late angiogenic stages. The process of angiogenesis involves alternating cycles of temporally and spacially regulated ECM synthesis and degradation. Proteins of the ECM can be deposited relatively early in angiogenesis e.g. fibronectin, as well as late in the differentiation phase e.g. collagen IV. Among the ECM constituents collagens, are associated best with in vivo angiogenesis. | ||
cytoskeletal remodeling & related signaling
| During the step of chemotactic migration endothelial cells repeatedly extend flexible protrusions from the cell body. The long filopodial-like cellular processes migrate along extracellular matrix components into the interstitial space. The listed TEMs are potential key participants in the signaling pathways that feedback from integrins to modulate the cytoskeleton. Additionally they might signal to the nucleus, via Rho family or MAPK pathways. The sequence analysis of TEM5, a RhoGEF, points at a potential link to MAPK signaling (possibly JNK). TEM24 has been implicated in JNK/SAPK regulation as well. Additionally Rho proteins, Rac and Cdc42, are known to regulate the activity of the c-Jun amino-terminal kinase (JNK). There is limited information on how G proteins are activated by cell surface receptors, as GPCR,cytokine and TGFbeta receptors. Interestingly G proteins and MAPKs, are also involved in the downstream signaling of TGF-beta family members besides Smad(TEM39). | ||
adhesion
| Integrins as the main adhesion and migration receptors of cells provide the information about the surrounding environment and regulate cell migration. TEM17 is a potential type I membrane protein with similarity to protein-families which have been implicated in axon migration. Thy-1 (TEM13) is a GPI-anchored protein having a role in the regulation of cell-cell adhesion and differentiation. | ||
endo- and exocytosis
| Endocytic receptors as uPARAP have been implicated in the process of focused proteolysis (uPAR-system). TEM1 might function in the internalization of a yet unknown ligand potentially supplied by blood cells. Peanut (TEM46) is a septin with a potential positive and/or a negative role in exocytosis. | ||
| From the 46 TEM SAGE-tags |  |
|
 |
| |
 |
|
|
 |
| |
|
||
| ||
|